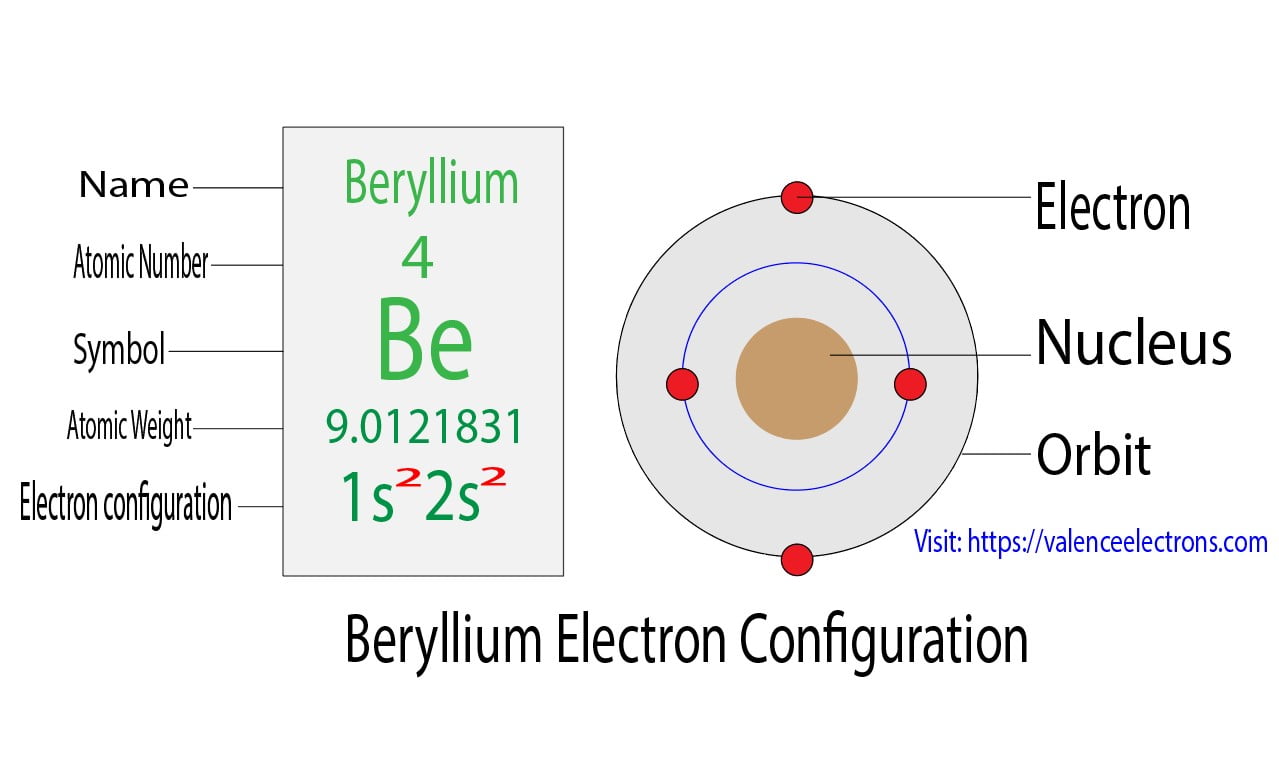
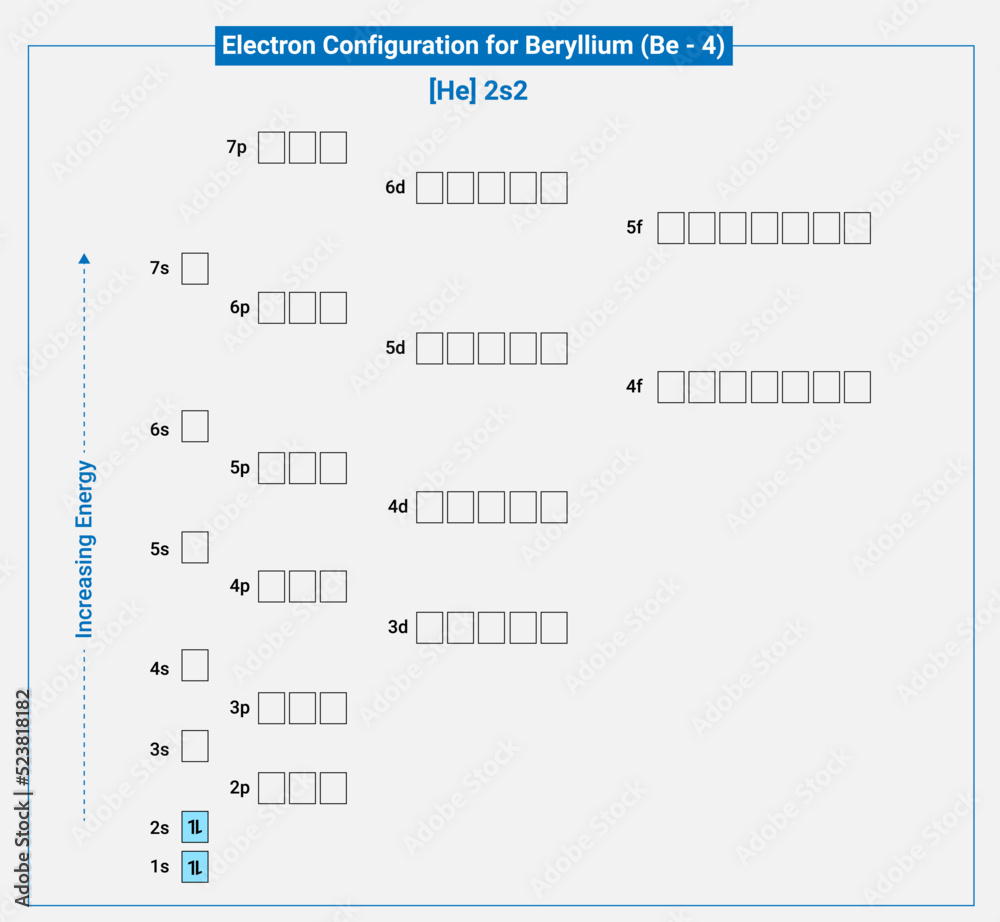
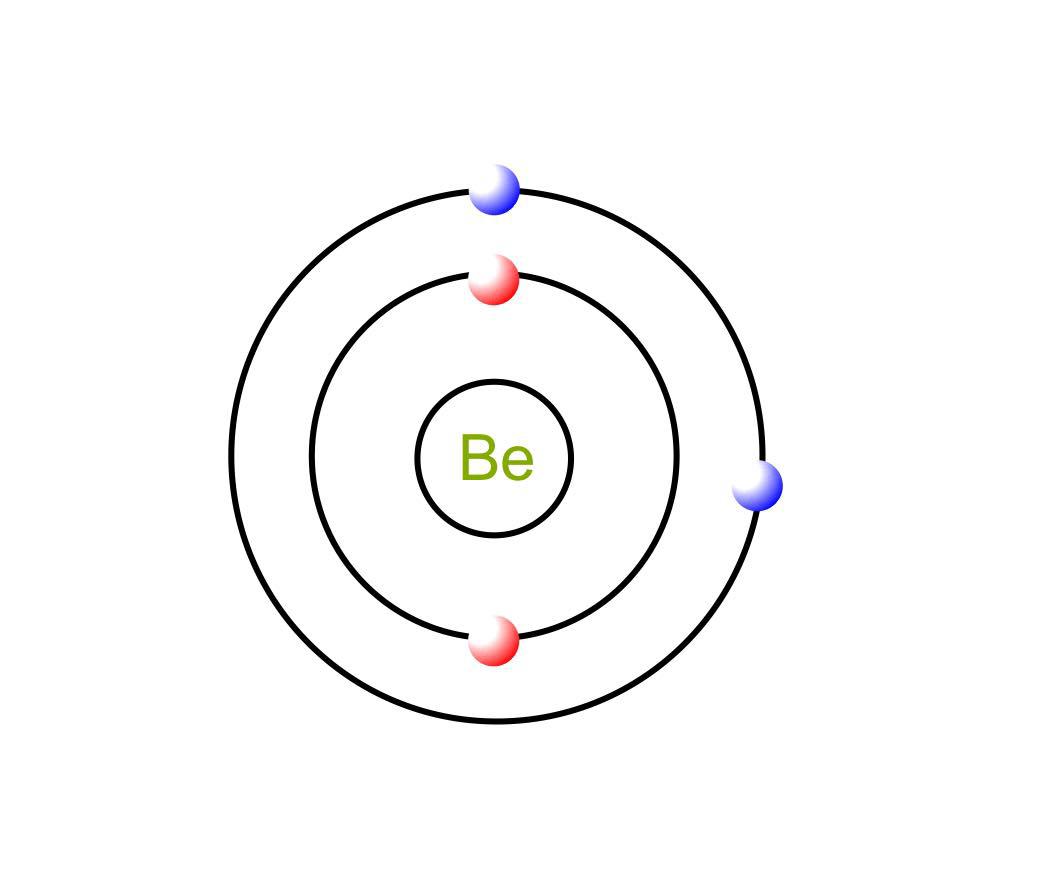
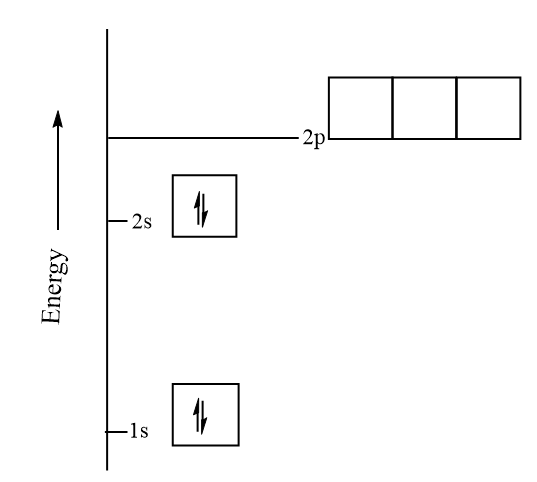
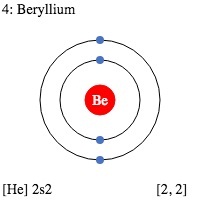
1. Indicate the number of unpaired electrons present in a beryllium atom. Is this atom paramagnetic or diamagnetic? 2. Indicate the number of unpaired electrons present in a cobalt atom. Is this

Electronic configuration of Beryllium spdf Trick -Digital Kemistry | Electron configuration, Chemistry lessons, Chemistry

File:Electron shell 004 Beryllium.svg - Wikimedia Commons | Atom diagram, Electron configuration, Atom






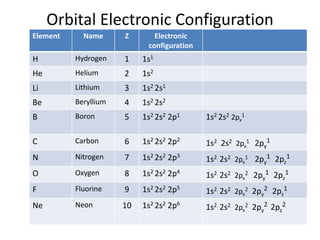
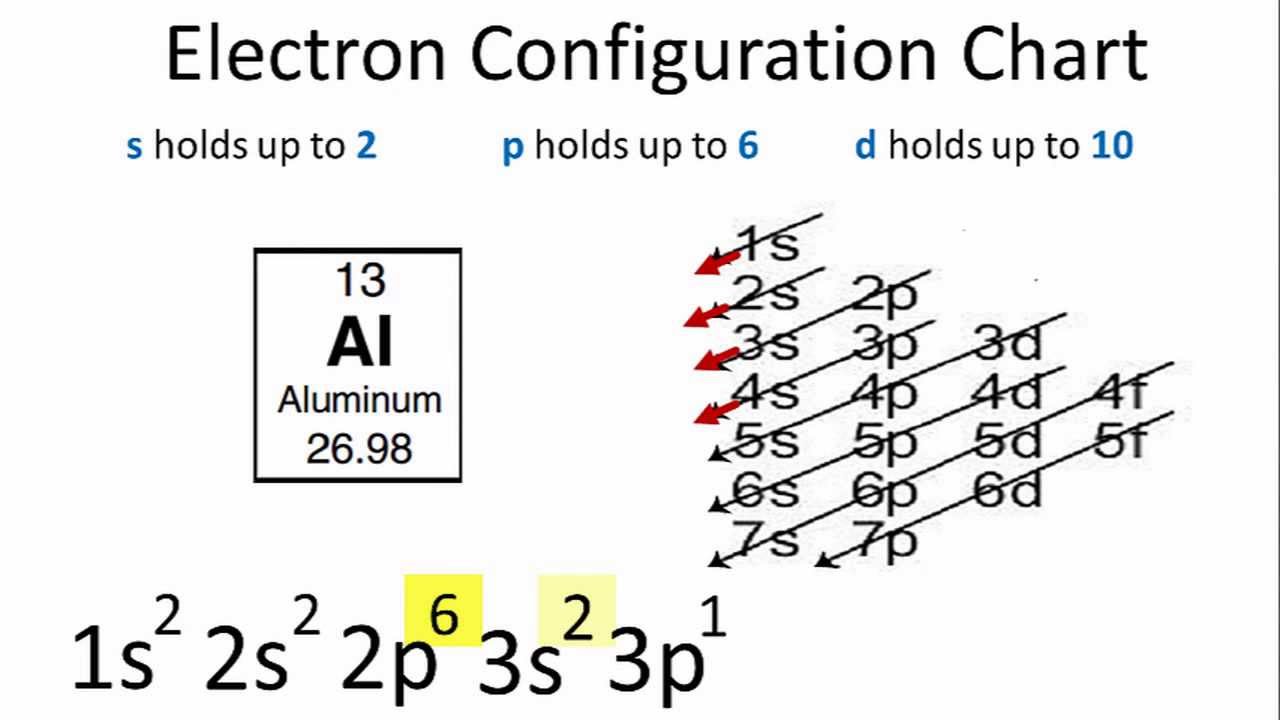




![ANSWERED] (1) Write the electron configuration for t... - Organic Chemistry - Kunduz ANSWERED] (1) Write the electron configuration for t... - Organic Chemistry - Kunduz](https://media.kunduz.com/media/sug-question/raw/54060479-1657532523.9837868.jpeg)